 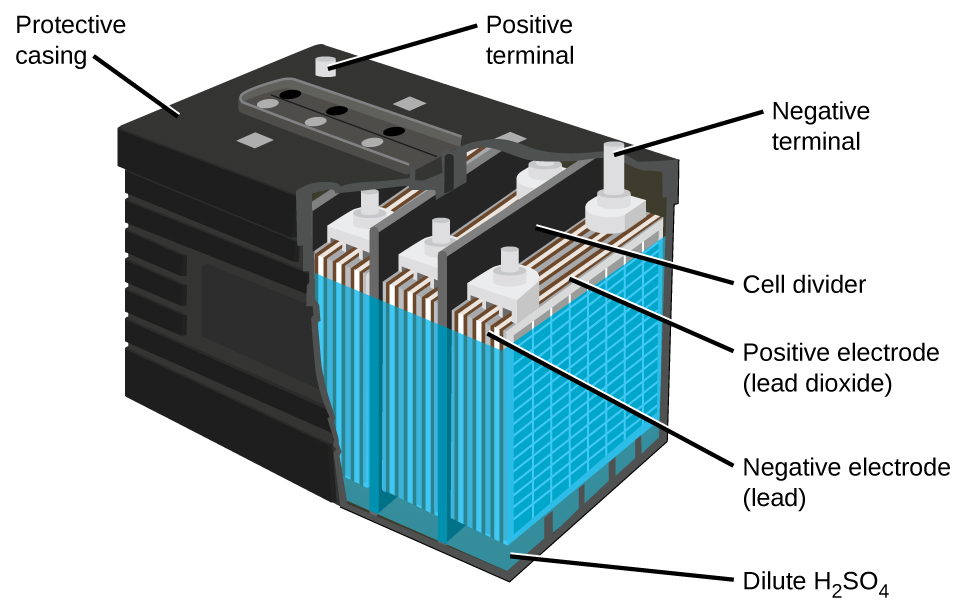 
of cell the cell works like an electrolytic cell and the electrode reactions get reversed. The cell can be recharged by passing electric current of a suitable voltage in the opposite direction. When the density of H 2SO 4 falls below 1.2 g mL -1, the battery needs recharging. As a result, the concentration of H 2SO 4 decreases and the density of the solution also decrease. It is clear from the above reactions that during the working of the cell, PbSO 4 is formed at each electrode and sulphuric acid is used up. The potential of the cell depends on the concentration (density) of H 2SO 4. Redox reaction in lead storage cell/ battery At the cathode PbO 2 is reduced to Pb 2+ ions and PbSO 4 is formed. Lead storage batteryĪt the anode, lead is oxidized to Pb 2+ ions and insoluble PbSO 4 is formed. The electrolyte is aqueous solution of sulphuric acid (38% by mass) having a density 1.30 g mL -1 sulphuric acid. In each cell, the anode is a grid of lead packed with finely divided spongy lead and the cathode is a grid of lead packed with PbO 2. Three to six such cells are generally combined to get 6 to 12 volt battery. Each battery consists of a number of voltaic cells connected in series. This is the most commonly used battery in automobiles. The cells which can be recharged and can be used again and again as a source of electric current are called secondary cells.įor example lead storage battery which is used in automobiles, Ni-Cd battery and Li ion battery. You can also watch the full video on this topic from the link at the bottom. In this post you will learn the working of lead storage battery which falls in the category of secondary cells. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
